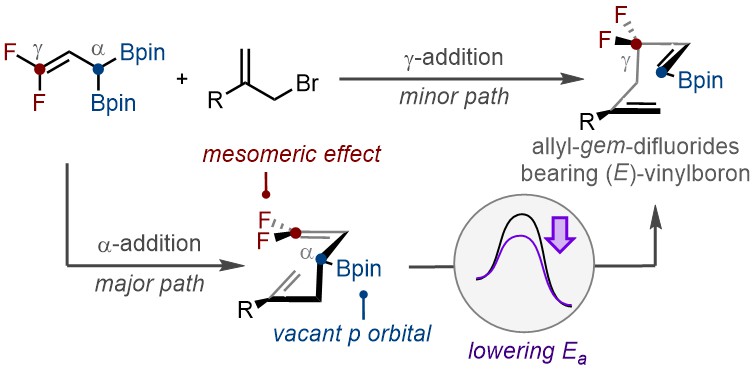
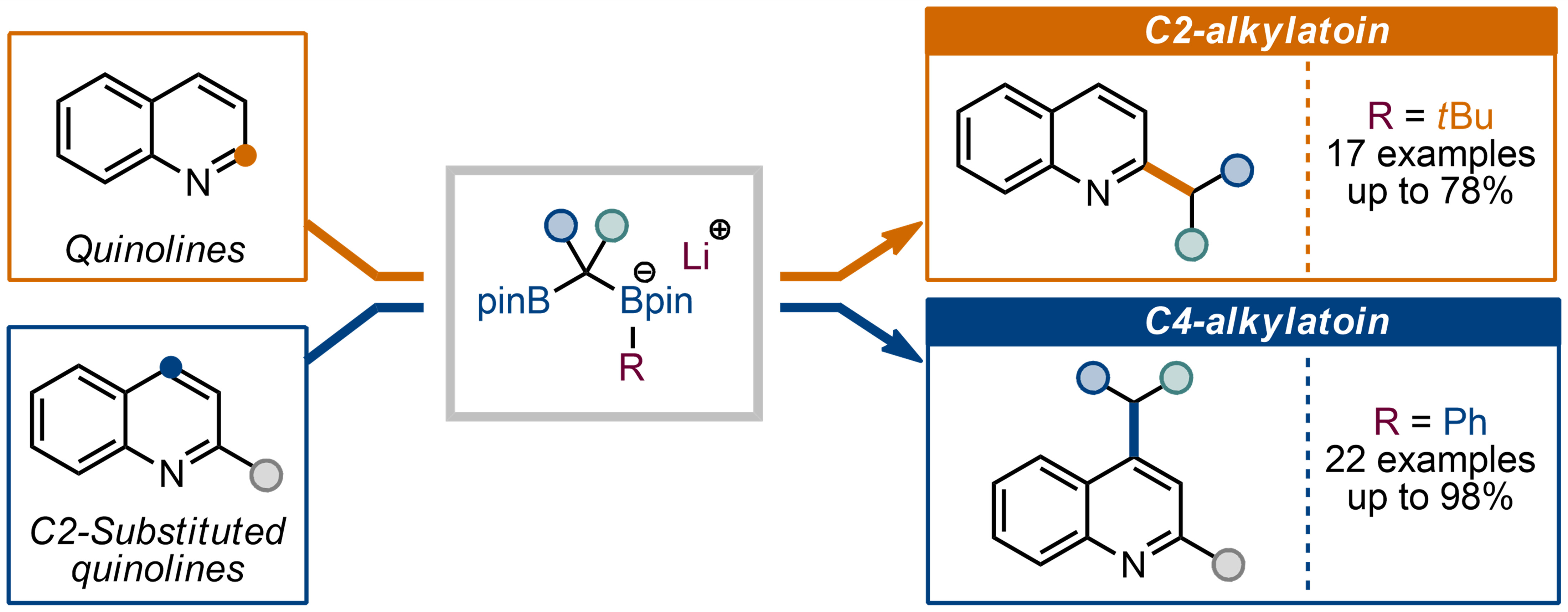
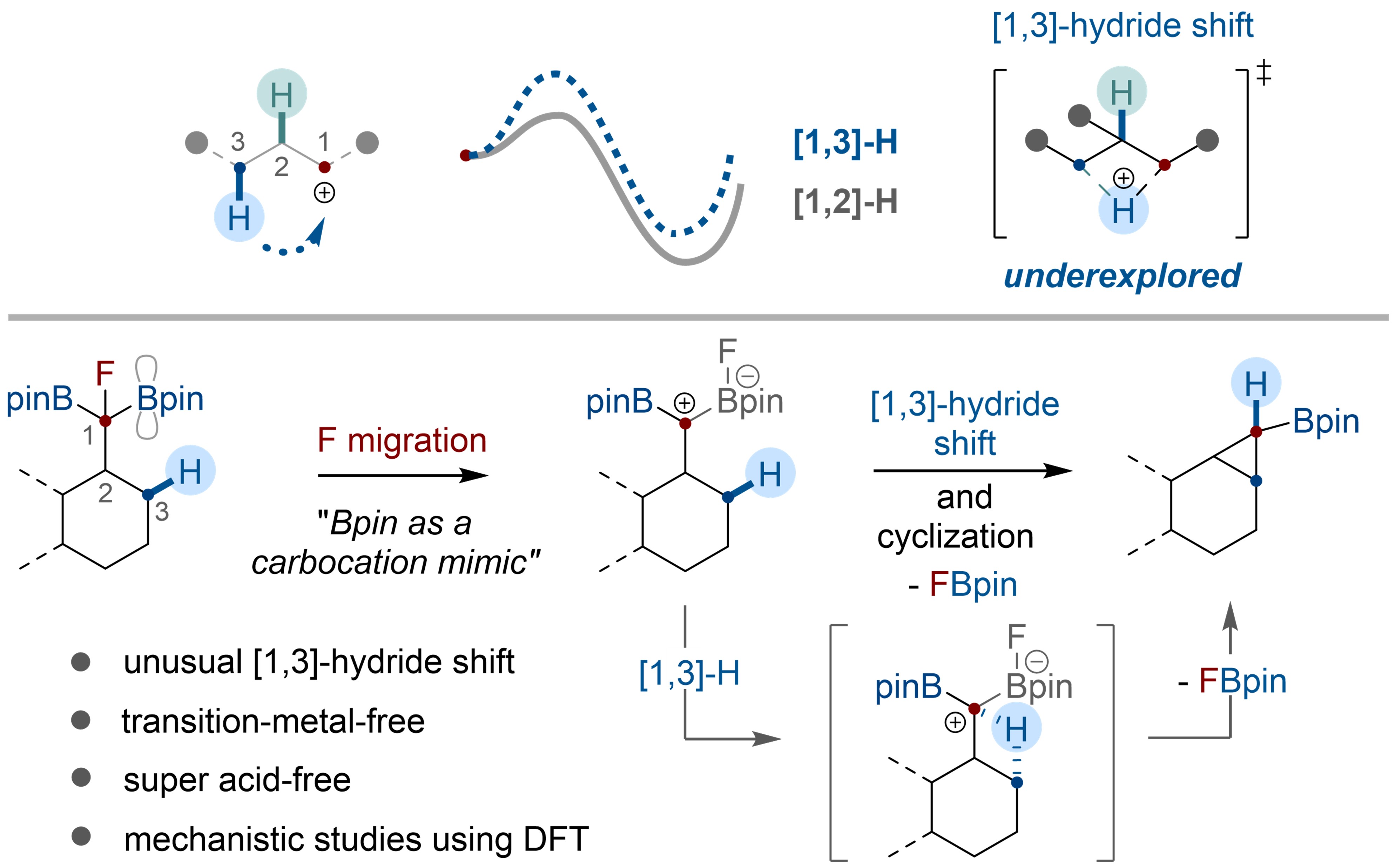
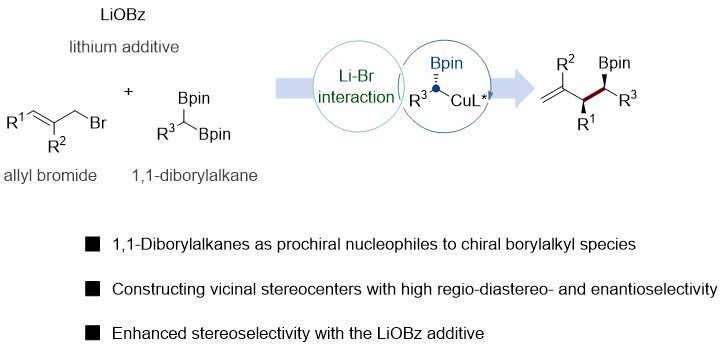
THE CHO
RESEARCH GROUP
Catalyst & Organic Synthesis Lab.
THE CHO
RESEARCH GROUP
Catalyst & Organic Synthesis Lab.
THE CHO
RESEARCH GROUP
Catalyst & Organic Synthesis Lab.
THE CHO
RESEARCH GROUP
Catalyst & Organic Synthesis Lab.